TuckerD
Well-Known Member
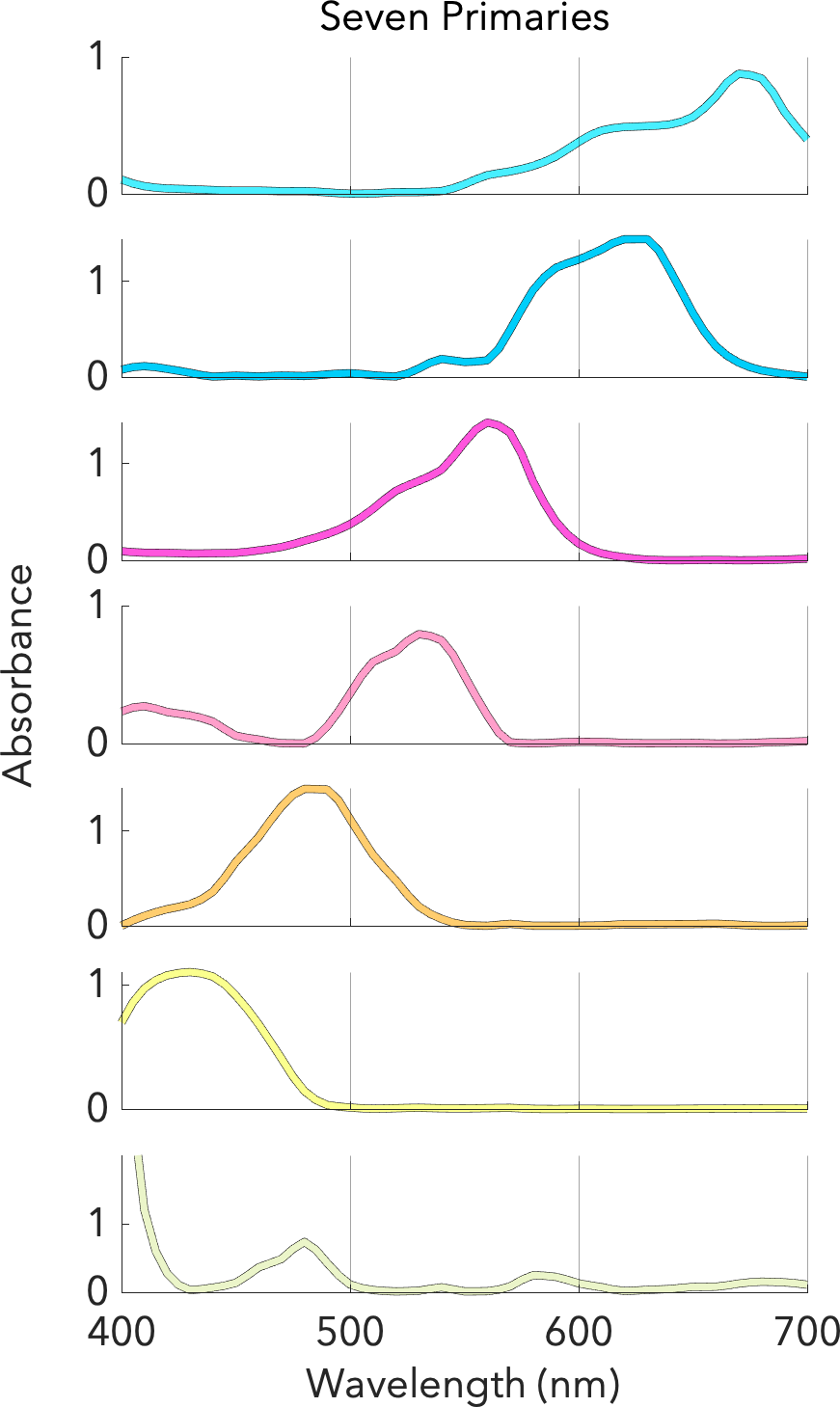
Hi all, I finally got around to writing up a project I did a few months ago. I wondered if I could apply spectral analysis techniques on the transmission curves of gels to figure out what pigments get mixed together. I originally wanted to measure Rosco gels on the spectrophotometer at my lab (Thanks for the gel book @dvsDave) but we are shut down for covid. So I used some LEE data I was able to gather instead. I'd love to share this work with you and get your feedback.
 tuckerd.info
tuckerd.info

Method of Spectral Optimization to Discover the Underlying Pigments in a Selection of Color Filters
Several months ago, as part of a term project in my graduate program, I was looking for a challenging problem, in a “new” area that I previously haven’t really worked in. And I wa…
 tuckerd.info
tuckerd.info